Abstract
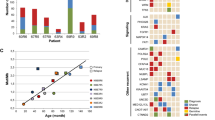
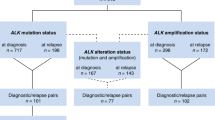
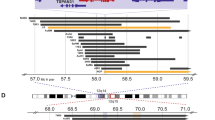
The majority of patients with neuroblastoma have tumors that initially respond to chemotherapy, but a large proportion will experience therapy-resistant relapses. The molecular basis of this aggressive phenotype is unknown. Whole-genome sequencing of 23 paired diagnostic and relapse neuroblastomas showed clonal evolution from the diagnostic tumor, with a median of 29 somatic mutations unique to the relapse sample. Eighteen of the 23 relapse tumors (78%) showed mutations predicted to activate the RAS-MAPK pathway. Seven of these events were detected only in the relapse tumor, whereas the others showed clonal enrichment. In neuroblastoma cell lines, we also detected a high frequency of activating mutations in the RAS-MAPK pathway (11/18; 61%), and these lesions predicted sensitivity to MEK inhibition in vitro and in vivo. Our findings provide a rationale for genetic characterization of relapse neuroblastomas and show that RAS-MAPK pathway mutations may function as a biomarker for new therapeutic approaches to refractory disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Change history
20 July 2015
In the version of this article initially published online, affiliation 30 stating that "These authors jointly supervised this work" was incorrectly omitted for author Jan J. Molenaar. The error has been corrected for the print, PDF and HTML versions of this article.
References
Hero, B. et al. Localized infant neuroblastomas often show spontaneous regression: results of the prospective trials NB95-S and NB97. J. Clin. Oncol. 26, 1504–1510 (2008).
Park, J.R. et al. Children's Oncology Group's 2013 blueprint for research: neuroblastoma. Pediatr. Blood Cancer 60, 985–993 (2013).
Simon, T. et al. Treatment and outcomes of patients with relapsed, high-risk neuroblastoma: results of German trials. Pediatr. Blood Cancer 56, 578–583 (2011).
Maris, J.M. Recent advances in neuroblastoma. N. Engl. J. Med. 362, 2202–2211 (2010).
Cohn, S.L. et al. The International Neuroblastoma Risk Group (INRG) classification system: an INRG Task Force report. J. Clin. Oncol. 27, 289–297 (2009).
Bown, N. et al. Gain of chromosome arm 17q and adverse outcome in patients with neuroblastoma. N. Engl. J. Med. 340, 1954–1961 (1999).
Attiyeh, E.F. et al. Chromosome 1p and 11q deletions and outcome in neuroblastoma. N. Engl. J. Med. 353, 2243–2253 (2005).
Seeger, R.C. et al. Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N. Engl. J. Med. 313, 1111–1116 (1985).
Caron, H. et al. Allelic loss of chromosome 1p as a predictor of unfavorable outcome in patients with neuroblastoma. N. Engl. J. Med. 334, 225–230 (1996).
Cheung, N.K. et al. Association of age at diagnosis and genetic mutations in patients with neuroblastoma. J. Am. Med. Assoc. 307, 1062–1071 (2012).
Molenaar, J.J. et al. Sequencing of neuroblastoma identifies chromothripsis and defects in neuritogenesis genes. Nature 483, 589–593 (2012).
Pugh, T.J. et al. The genetic landscape of high-risk neuroblastoma. Nat. Genet. 45, 279–284 (2013).
Sausen, M. et al. Integrated genomic analyses identify ARID1A and ARID1B alterations in the childhood cancer neuroblastoma. Nat. Genet. 45, 12–17 (2013).
Taggart, D., Dubois, S. & Matthay, K.K. Radiolabeled metaiodobenzylguanidine for imaging and therapy of neuroblastoma. Q. J. Nucl. Med. Mol. Imaging 52, 403–418 (2008).
Schleiermacher, G. et al. Emergence of new ALK mutations at relapse of neuroblastoma. J. Clin. Oncol. 32, 2727–2734 (2014).
Carter, S.L. et al. Absolute quantification of somatic DNA alterations in human cancer. Nat. Biotechnol. 30, 413–421 (2012).
Boca, S.M., Kinzler, K.W., Velculescu, V.E., Vogelstein, B. & Parmigiani, G. Patient-oriented gene set analysis for cancer mutation data. Genome Biol. 11, R112 (2010).
Futreal, P.A. et al. A census of human cancer genes. Nat. Rev. Cancer 4, 177–183 (2004).
Bresler, S.C. et al. ALK mutations confer differential oncogenic activation and sensitivity to ALK inhibition therapy in neuroblastoma. Cancer Cell 26, 682–694 (2014).
Hölzel, M. et al. NF1 is a tumor suppressor in neuroblastoma that determines retinoic acid response and disease outcome. Cell 142, 218–229 (2010).
Tartaglia, M. et al. Diversity and functional consequences of germline and somatic PTPN11 mutations in human disease. Am. J. Hum. Genet. 78, 279–290 (2006).
Carr-Wilkinson, J. et al. High frequency of p53/MDM2/p14ARF pathway abnormalities in relapsed neuroblastoma. Clin. Cancer Res. 16, 1108–1118 (2010).
Garnett, M.J. et al. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature 483, 570–575 (2012).
Schleiermacher, G. et al. Accumulation of segmental alterations determines progression in neuroblastoma. J. Clin. Oncol. 28, 3122–3130 (2010).
Landau, D.A. et al. Evolution and impact of subclonal mutations in chronic lymphocytic leukemia. Cell 152, 714–726 (2013).
Long, G.V. Increased MAPK reactivation in early resistance to dabrafenib/trametinib combination therapy of BRAF-mutant metastatic melanoma. Nat. Commun. 5, 5694 (2014).
George, R.E. et al. Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature 455, 975–978 (2008).
Mossé, Y.P. et al. Safety and activity of crizotinib for paediatric patients with refractory solid tumours or anaplastic large-cell lymphoma: a Children's Oncology Group phase 1 consortium study. Lancet Oncol. 14, 472–480 (2013).
Berry, T. et al. The ALKF1174L mutation potentiates the oncogenic activity of MYCN in neuroblastoma. Cancer Cell 22, 117–130 (2012).
Moore, N.F. et al. Molecular rationale for the use of PI3K/AKT/mTOR pathway inhibitors in combination with crizotinib in ALK-mutated neuroblastoma. Oncotarget 5, 8737–8749 (2014).
Drmanac, R. et al. Human genome sequencing using unchained base reads on self-assembling DNA nanoarrays. Science 327, 78–81 (2010).
Favero, F. et al. Sequenza: allele-specific copy number and mutation profiles from tumor sequencing data. Ann. Oncol. 26, 64–70 (2015).
Krzywinski, M. et al. Circos: an information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Cibulskis, K. et al. Sensitive detection of somatic point mutations in impure and heterogeneous cancer samples. Nat. Biotechnol. 31, 213–219 (2013).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012).
Rausch, T. et al. DELLY: structural variant discovery by integrated paired-end and split-read analysis. Bioinformatics 28, i333–i339 (2012).
Carter, S.L. et al. Absolute quantification of somatic DNA alterations in human cancer. Nat. Biotechnol. 30, 413–421 (2012).
Roth, A. et al. PyClone: statistical inference of clonal population structure in cancer. Nat. Methods 11, 396–398 (2014).
Lamers, F. et al. Targeted BIRC5 silencing using YM155 causes cell death in neuroblastoma cells with low ABCB1 expression. Eur. J. Cancer 48, 763–771 (2012).
Gnirke, A. et al. Solution hybrid selection with ultra-long oligonucleotides for massively parallel targeted sequencing. Nat. Biotechnol. 27, 182–189 (2009).
Acknowledgements
The authors would like to thank N. Ross for sample preparation and quality control. This study was supported in part by US National Institutes of Health grants RC1MD004418 to the TARGET consortium and CA98543 and CA98413 to the Children's Oncology Group and was supported in part by a grant throughout the University of Pennsylvania Genome Frontiers Institute. In addition, this project was funded in part with federal funds from the National Cancer Institute, US National Institutes of Health, under contract HHSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the US Department of Health and Human Services nor does mention of trade names, commercial products or organizations imply endorsement by the US government. The binimetinib xenograft studies were supported through a research collaborative agreement between the Children's Hospital of Philadelphia and Novartis Pharmaceuticals. In France, this study was supported by the Annenberg Foundation and the Nelia and Amadeo Barletta Foundation. Funding was also obtained from SiRIC/INCa (grant INCa-DGOS-4654) and from the Comité d'Evaluation et Suivi des Projets de Recherche de Transfert (CEST) of Institut Curie. This study was also funded by the Associations Enfants et Santé, Association Hubert Gouin Enfance et Cancer, Les Bagouz à Manon and Les Amis de Claire. Deep sequencing experiments were conducted on the ICGex next-generation sequencing platform at the Institut Curie funded by the EQUIPEX Investissements d'Avenir program (ANR-10-EQPX-03) and ANR10-INBS-09-08 from the Agence Nationale de le Recherche and by the Canceropôle Ile-de-France. In the Netherlands, this study was supported by grants from the Villa Joep Foundation, the Kinderen Kankervrij Foundation and the Netherlands Cancer Foundation.
Author information
Authors and Affiliations
Contributions
J.M.M., J.J.M. and G.S. co-coordinated the project. T.F.E., D.A.O., V.B., J. Koster, R.V., G.S., J.J.M. and J.M.M. prepared the manuscript. D.A.O., J. Koster, L.C.D., V.B., N.B.B., P.L.-N., S.J.D., T.J.P., E.F.A., J.S.W., S.Z., S.A., T.B.K.W., D.A.Z. and A.N. generated microarray and/or next-generation sequencing data and/or performed computational data analysis. T.F.E., L.S.H., J.R. and J.M.M. conceived drug testing and gene manipulation experiments and analyzed the data. P.v.S., M.E.E., E.L., V.C., A.B. and M.C. performed sample preparation. L.S.H., J.R., L.S., A.H., M.E.M.D. and T.F.E. contributed to in vitro drug testing and manipulation experiments. A.B., M.C., P.L.-N., D.A.O., T.J.P., E.v.W. and C.E.v.d.S. performed subclonal detection of mutational and structural variants. J.M.G.-F., M.D.H., E.M.W., J.H.S., G.A.T., H.N.C., J.M. and V.C. contributed patient tissue and/or other biological reagents. M.A.S., J.M.G.A., D.S.G., J. Khan and J.M.M. organized the US neuroblastoma TARGET consortium sequencing effort. J.J.M., R.V., J. Koster. and H.N.C. organized the Dutch consortium sequencing effort. G.S., J.M., I.J.-L. and O.D. organized the French consortium sequencing effort.
Corresponding authors
Ethics declarations
Competing interests
J.M.M. has commercial research grants from Novartis Pharmaceuticals and GlaxoSmithKline. H.N.C. is employed by Roche.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17 and Supplementary Note. (PDF 10668 kb)
Supplementary Tables 1–6
Supplementary Tables 1–6. (XLSX 393 kb)
Rights and permissions
About this article
Cite this article
Eleveld, T., Oldridge, D., Bernard, V. et al. Relapsed neuroblastomas show frequent RAS-MAPK pathway mutations. Nat Genet 47, 864–871 (2015). https://doi.org/10.1038/ng.3333
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3333
This article is cited by
-
The RAS oncogene in brain tumors and the involvement of let-7 microRNA
Molecular Biology Reports (2024)
-
Effective methods for bulk RNA-seq deconvolution using scnRNA-seq transcriptomes
Genome Biology (2023)
-
A CRISPR-drug perturbational map for identifying compounds to combine with commonly used chemotherapeutics
Nature Communications (2023)
-
Lorlatinib with or without chemotherapy in ALK-driven refractory/relapsed neuroblastoma: phase 1 trial results
Nature Medicine (2023)
-
Clonal evolution during metastatic spread in high-risk neuroblastoma
Nature Genetics (2023)